
One of the main goals in every industry whether in manufacturing or in service, is to have a smooth and reliable operation of every equipment or machine used.
One way to achieve this goal is to implement a quality control procedure, a Preventive Maintenance.
In a calibration, preventive maintenance is also belonging to an activity we called Intermediate Check.
In this article, I will present the process of preventive maintenance in a calibration lab. This can be implemented whether in an In-House Calibration or a 3rd party Calibration Lab
I will discuss here the requirements under the standard ISO 17025:2017 relating to preventive maintenance or PM, advantages of preventive maintenance in calibration, PM schedules, the content of a PM Checklist and PM procedure.
What is Preventive Maintenance?
Preventive maintenance or PM is a familiar term mostly related to equipment maintenance. This term is often heard when you are a technician.
Below are the definitions of preventive maintenance as defined by WIKIPEDIA.
- The care and servicing by personnel for the purpose of maintaining equipment in satisfactory operating condition by providing for systematic inspection, detection, and correction of incipient failures either before they occur or before they develop into major defects.
- The work carried out on equipment in order to avoid its breakdown or malfunction. It is a regular and routine action taken on equipment in order to prevent its breakdown.
- Maintenance, including tests, measurements, adjustments, parts replacement, and cleaning, performed specifically to prevent faults from occurring.
.
Preventive Maintenance in Calibration
Why do we perform Preventive Maintenance (PM)?
The definitions that are provided above clearly state the reason why we perform a PM. An additional reason is that it is a requirement under a recognized international standard.
By performing preventive maintenance, we are fulfilling the below requirements under ISO 17025:2017.
The requirements are under these clauses:
Clause 6.4 Equipment:
6.4.3 The Laboratory shall have a procedure for handling, transport, storage, use, and planned maintenance of equipment in order to ensure the proper functioning and to prevent contamination or deterioration.
6.4.10 when intermediate checks are necessary to maintain confidence in the performance of the equipment, these checks shall be carried out according to a procedure.
Clause 7.7 Ensuring the Validity of results:
7.7.1 The laboratory shall have a procedure for monitoring the validity of results. The resulting data shall be recorded in such a way that the trends are detectable and, where practicable, statistical techniques shall be applied to review the results. This monitoring shall be planned and reviewed and shall include, where appropriate, but not limited to:
- functional checks of measuring and testing equipment;
- Intermediate checks on measuring equipment;
Based on the requirements above from ISO 17025, we should have a procedure in place that is planned maintenance in order to:
- ensure proper functioning of our equipment;
- maintain confidence in the performance of our equipment
- ensuring the validity of results through functional checks and intermediate checks on our measuring equipment commonly known as the reference standard.
These requirements can be fulfilled through the process of Preventive Maintenance.
Advantages of Preventive Maintenance in the Calibration Process
- Ensures proper functioning of reference standards
- Fulfilling the requirement of ISO standards like ‘ensuring the validity of results’ and ‘Intermediate check.’
- Once data is recorded, you will have a tracking record of its performance, thus, a good way to justify an extension for a calibration interval.
- Prolongs the life of your reference standard.
- You are confident that your reference standards are providing the best measurement output.
.
Preventive Maintenance (PM) Procedure
PM is a process because it involves a combination of activities or steps, not just directly performing maintenance. We are following a Preventive Maintenance Program or a PM procedure to execute properly.
The requirements are:
- Determine what equipment or instruments need PM
- Define a schedule of execution, a PM plan – how often should we perform a preventive maintenance
- Create a Preventive Maintenance checklist – the technical content
- Performing preventive maintenance – Involves functionality check and partial calibration
- Labeling through a PM logFile
- Reports submission, approval, and filing
With these requirements, we can now define a Preventive Maintenance Procedure in order to standardize and document it as part of our quality system.
1. Determine what instruments or equipment that needs preventive maintenance
All standards that are used for calibration or testing should be included in the PM list. Preventive Maintenance does not necessarily mean that it is only for reference standards that are with mechanical or electrical parts, it can include those that are for cleaning only. An example of this is the gauge blocks and the standard weights.
Our primary objective for performing PM is to have a system in place that will ensure each of our reference standards are in good condition while it is under the provided calibration interval.
2. Define Schedule of PM
There is no standard set about PM execution. This is based on every need, manufacturer’s recommendation, and design of every equipment or laboratory. The important thing is we should set a defined frequency in performing a PM.
This defined frequency is scheduled by using a PM calendar. A simple excel sheet can do the job.
In the lab that I worked for, we scheduled our PM every 3 months. Since our equipment is reference standards used for calibration, it is a good choice to have them scheduled more frequently to ensure that they are in confidence all the time.
3. Create a Preventive Maintenance Checklist
With all the parts and details in reference standards (equipment), it is a must that we should have a form called a PM checklist.
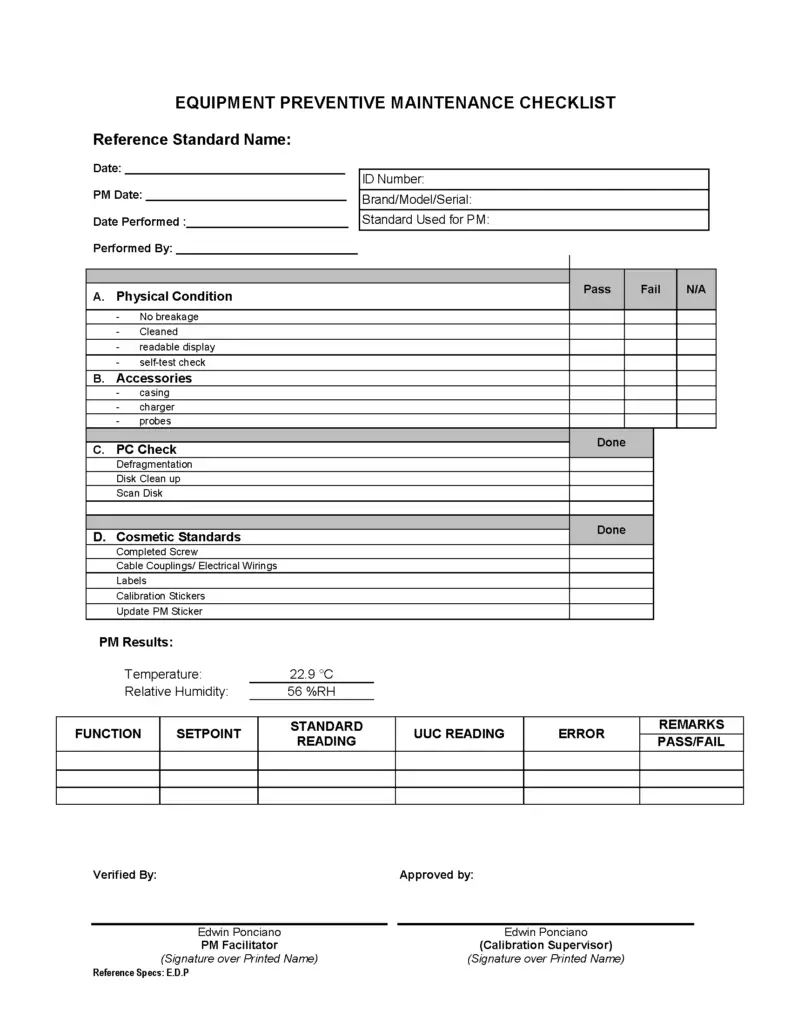
PM checklist will simplify our work and will provide a continuous flow of maintenance activity. It is a step by step list to be accomplished. see image
The checklist also serves as our preventive maintenance report.
The content on a checklist includes:
- Details of the reference standards ( make, model, serial #, etc)
- PM date;
- PM due;
- PM by;
- Reviewed by;
- Functionality check
- Accessories check;
- Labels check- sticker and seals;
- Calibration
- Others – depends on the design and requirements of the equipment
4. Performing the Maintenance
Below are some of the most important steps in the PM procedure
- Cleaning
- Assessing, checking, and recording the status of physical condition- some details to be checked are the labels, stickers, and seals if it is still complete and readable.
- Functionality check – involves the check for battery status, power-up conditions, readable display, functional button, and many more.
- Intermediate check or partial calibration – we will perform a simple calibration in order to verify the accuracy of its reading.
- Labeling – this is by accomplishing a Preventive Maintenance Log for proper monitoring and serves as a history file.
5. PM Reports Approval and Filing
Once PM is done, PM reports are submitted for approval and recording. This is one of the most time-consuming parts in PM activities, to update a record and analyze the data. This is where statistical techniques will be applied. An example is by using the control chart.
PM records will be included and filed under Technical Records, therefore, it should follow the records control procedure designed for every records made.
.
Conclusion
Preventive Maintenance is one of the major activities inside a laboratory that is integrated into our quality management system that guides our everyday operation. In this post, I have presented the following:
- Requirements for quality control under ISO 17025 as part of the quality system procedure.
- Preventive Maintenance (PM) Procedure which includes:
- PM schedule
- The PM Checklist and its contents
- Steps of PM execution
- PM reports and approval
I hope that you are now planning on creating a PM plan if you do not have one.
Thank you for visiting, please comment and subscribe
Best regards,
edwin
14 Responses
Vinod
Thanks for your post, please provide a quality manual as per ISO 17025:2017, we are unable to create it.
edsponce
Hi Mr. Vinod,
You are welcome.
If you are now using a 2017 version of the ISO 17025, a quality manual is already not a requirement.
As long as you can provide a documented proof that you complied to all the necessary requirements, then there is no need for a quality manual.
Best regards,
Edwin
Islam
Very good article
Thank you
edsponce
I appreciate you liked it. thank you for the comment.
Best regards,
Edwin
Vinod
Hi Edwin, thanks for your post. Can you differentiate intermediate checks and functionality check?
Is recalibration requirement is there in the standard, if yes then how to do it and what should be frequency of recalibration.
In equipment history card what details should be logged. Please respond
edsponce
Hi Vinod,
You are welcome.
An intermediate check is a process or planned activity that is part of a quality control procedure to ensure the confidence of a reference standard. Ensuring confidence by doing verification of readings at a chosen range. During the Intermediate check, we are performing functionality check and verification or simply functionality check.
Functionality check is a procedure wherein you will check not just the displayed measurement result but including physical observations like self-test, power-on test, the functionality of buttons and others. Depending on your format, part of the functionality check is the verification, to check if the readings are within accepted tolerance.
The main difference between the two is that intermediate check is a process performed at planned date or interval while functionality check is a procedure within the Intermediate check or part of the intermediate check process.
Examples of activities where we implement intermediate checks are Preventive Maintenance and onsite calibration.
I have explained in detail about intermediate check in my other post in this link.
Equipment history card is usually used during Preventive Maintenance. It is a card that is placed for monitoring purposes and to display its status. As a minimum, below are my suggestion for its content:
1. Details of the standards like manufacturer, model, serial#. ID#
2. Date performed
3. Performed by
4. Checked by
5. Remarks: Pass/fail
Regarding recalibration, yes, recalibration is a requirement. The reason why we “calibrate” also applies to why we “recalibrate”. Below are some of the related clauses from ISO 17025:2017 that presented and requiring recalibration, these clauses are:
1. 6.4.7 The laboratory shall establish a calibration program, which shall be reviewed and adjusted as necessary in order to maintain confidence in the status of calibration.
2. 7.7.1 The laboratory shall have a procedure for monitoring the validity of results. g) retesting or recalibration of retained items;
3. 6.4.13 Records shall be retained for equipment that can influence laboratory activities. The records shall include the following, where applicable: e) calibration dates, results of calibrations, adjustments, acceptance criteria, and the due date of the next calibration or the calibration interval;
As per ISO 17025, there are no requirements for the frequency of recalibration, the decision is up to the user, and these decisions could be based on his experience, based on the recorded performance or performance history of the standard and recommendations of the manufacturers.
Some accreditation bodies have a requirement so better ask them if you applied for accreditation.
There are no recalibration frequency requirements but there are guides that give recommendations, you may check “OIML D 10:2007 Guidelines for the determination of calibration intervals of measuring instruments” or other equivalent reference guides.
I hope this helps. If you have more concerns, do not hesitate to comment further.
Edwin
M. Saleh
Eng. Edwin,
In the context of your presntation, what is the role of ISO 10012?
Thanks in advance.
edsponce
Hi M. Saleh,
I am not implementing ISO 10012. Please elaborate more your question.
Best regards,
Edwin
Joseph Rindone
Though not a consideration of 17025 is not a quality manual something that one might need to satisfy the 9000 series?
edsponce
Hi Sir,
It is a good way to satisfy the requirements for monitoring and ensuring valid results.
Thank you for reading my posts.
Edwin
R. BELTRAN
Hello Edwin-
Is it necessary to be lab certified to an accredited organization if calibrations are conducted with a PM?
edsponce
Hi R. Beltran,
It is not necessary, you just need to maintain a record of the traceability of the reference standard you use and a competency evidence of the personnel involve (training record).
Accreditations are only for calibration labs that provide 3rd party calibration services.
I hope this helps,
Edwin
Josphat
Good presentation. Looking forward to learning more.
edsponce
Hi Josphat,
Thanks for reading my posts.
Best regards,
Edwin