
Calibration, verification, and validation are always used or read in every measurement related topic.
When I was just starting out in this calibration environment, I was always confused on how or when to use each term.
There were so many helpful articles that I read and review, and still afterward, when I needed to use each term, sometimes I still get confused.
I believe this also happens to most of you out there, whether reading or understanding a certain procedure or instructions.
In this post, I will present with you the:
- differences between each term,
- its applications in our measurement process (calibration Process)
- the ISO standards that require each to term to be performed
- some examples to illustrate each term
That I hope will help you to understand and interpret each requirement for managing our calibration process.
Definitions of Calibration, Verification, and Validation as per JCGM 200:2012
Below are the definitions that I directly copied from the International vocabulary of metrology or VIM for short.
Calibration
The operation that, under specified conditions, in a first step, establishes a relation between the quantity values with measurement uncertainties provided by measurement standards and corresponding indications with associated measurement uncertainties and, in a second step, use this information to establish a relation for obtaining a measurement result from an indication
Verification
provision of objective evidence that a given item fulfills specified requirements
Validation
verification, where the specified requirements are adequate for an intended use
Understanding the differences in Calibration, Verification, and Validation as applicable to Calibration Management
Calibration, verification, and validation are mostly used interchangeably if you are not aware of their differences and true meaning.
For the purpose of simplicity, I will focus this presentation in relation to measuring instruments as applied to the calibration process.
Given the definitions above, which is very technical, I will provide a more detailed and simple explanation in order to make it clear and more meaningful (I hope so), based on what I understand and experience in applying its concept in the calibration field.
Calibration
As with most simple definitions that I read, calibration is simply the “comparison” of the unknown reading of a UUC to a known reading of a Reference Standard, also known as the Master.
Based on a reference standard, you are comparing the performance of the unit under calibration (UUC). Through comparison, you can determine the accuracy and error or how far is the reading to the true value.

But the process does not end here, During calibration, we also consider:
- The traceability, the unbroken chain of comparison linking to the highest level (International standard like NIST)
- computed Measurement Uncertainty
Without these two, we cannot call it calibration; we are just performing a simple verification.
Also, in Calibration:
- you may or may not include adjustment or repair
- based on the result, pass or failed decisions are decided by the user as per their process acceptance criteria.
Your objective in performing calibration is guided by:
- customer requirements and regulatory authorities
- calibration procedures based on standard methods (from national, international or technical organizations)- also known as standard methods
- non-standard method – a validated laboratory-developed methods
- procedures from manufacturers or OEM
During calibration, we
- choose the required calibration procedure to be used
- compare to a reference standard (full-range or as per customer test point)
- determine the error
- adjust if capable
- check the traceability
- estimate measurement uncertainty
- record results to a calibration certificate
Verification
Verification is a process of “confirming” that a given specification is fulfilled.
It is a simple check to confirm that a certain instrument or equipment meets the intended output basing it to the requirements of certain specifications, not a comparison to a higher standard.
Your objective is guided by specifications from:
- a legal or regulatory body, examples are ASTM and ISO
- Manufacturer requirements.
- process or user requirements
Unlike Calibration where decisions are made based on how far or close the reading of the instrument in comparison to the provided results of the reference standard.

In verification, you are:
- Confirming the performance of the instrument based on given specifications or requirements.
- Ensuring that the instrument is working correctly for its intended purpose.
- Not checking the entire range. Just only to ensure that it is acceptable on its working or intended range
- Directly deciding what to do base on the verification result. These decisions are either:
- to recalibrate
- to adjust,
- extend usability
- scrap or make obsolete
- repair
Note that, we perform verification after calibration.
The activities where verification is involved in a calibration lab are:
- Intermediate check, which is used
- Before and after onsite calibration
- During the acceptance of newly purchased standards
- Once a reference standard has arrived from a third party calibration
- Preventive maintenance
- Receiving process of instruments for calibration
Validation
Validation is for “ensuring” the acceptability of the implemented measurement process.
It is the combined effect of calibration and verification but the final result is in the final output of the process.
The application in the calibration field is more on the development (design and development) of a calibration procedure in order to meet certain requirements.
A non-standard method can be used In a calibration laboratory, considering that it is validated before use.
During the validation of a calibration procedure, our objective is to determine:
- if the components introduced is capable for its intended use,
- relevant to customers’ needs
- consistent with the specifications of the instruments.
Another application of validation is qualifying a group of measuring devices that function as a system.
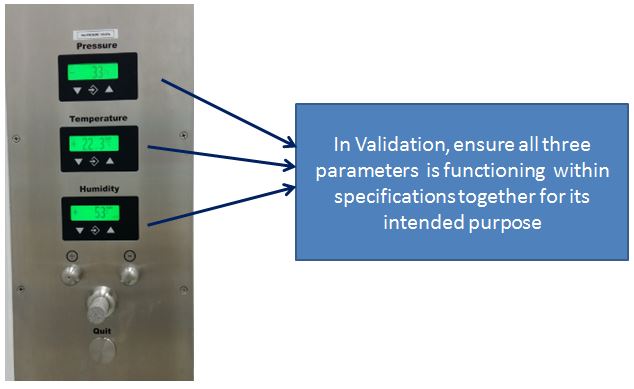
An example is a pressure transmitter, it consists of a pressure sensor and a transducer to convert a pressure signal to a current (4-20 mA) parameter in order to feed into a PLC display that performs a specific control and monitoring.
Moreover, we are focusing on validating the system as a whole, not just the instrument that is installed.
All parameters should be calibrated and verified before it can be used as a whole.
In Other words, In order to validate a process or procedure, you need to perform a calibration and verification.
The final output of validation is documented in a validation report where all results are recorded with a final decision for its suitability to be used in its intended purpose. It will also include the signatures of the persons who will approve the performed validation.
Techniques Used For Method Validation as per ISO 17025:
- calibration or evaluation of bias and precision using reference standards or reference materials;
- systematic assessment of the factors influencing the result;
- testing method robustness through variation of controlled parameters, such as incubator temperature, volume dispensed;
- comparison of results achieved with other validated methods;
- interlaboratory comparisons;
- the evaluation of measurement uncertainty of the results based on an understanding of the theoretical principles of the method and practical experience of the performance of the sampling or test method.
As per the above techniques, we can see that every validation technique is a process in itself.
For a specific example on how to use these terms, visit my other post in this link: How to differentiate calibration, verification, and validation?
You may also want to learn about the Differences Between Accuracy, Error, Tolerance, and Uncertainty in a Calibration Results
Why do we perform calibration, verification, and validation?
We are performing each activity to ensure quality in our measurement process and to comply as per the requirements of a standard.
These standards are ISO 17025 and ISO 9001. I will present the clauses where each term is used.
As per ISO 9001:2015
Measurement traceability under Clause 7.1.5.2
When measurement traceability is a requirement or is considered by the organization to be an essential part of providing confidence in the validity of measurement results, measuring equipment shall be:
- calibrated or verified, or both, at specified intervals, or prior to use, against measurement standards traceable to international or national measurement standards; when no such standards exist, the basis used for calibration or verification shall be retained as documented information;
>> The above requirements show that calibration and verification are used in ensuring measurement traceability or confidence in the validity of measurement results.
>> Validation will be performed when no such standard exists, and the basis of validation is through calibration and verification,
We can either perform calibration or verification or both.
AS per ISO 17025:2017
Under Clause 6.2.6
The laboratory shall authorize personnel to perform specific laboratory activities, including but not limited to, the following:
- a) development, modification, verification and validation of methods;
Under Clause 6.4.13
Records shall be retained for equipment that can influence laboratory activities. The records shall include the following, where applicable:
- c) evidence of verification that equipment conforms with specified requirements;
Under Clause 7.1.7
The laboratory shall cooperate with customers or their representatives in clarifying the customer’s request and in monitoring the laboratory’s performance in relation to the work performed.
Such cooperation includes the preparation, packaging, and dispatch of items needed by the customer for verification purposes.
Under Clause 7.2
Selection, verification, and validation of methods
>>As per above requirements, we can see that verification and validation are applicable in terms of:
- Assigning authorize personnel performing verification and validation of methods;
- Records should be maintained for verification and validation performed
- Lab cooperation for customer verification purposes as part of customer service
- Selection, verification, and validation of methods used in calibration or testing activities.
Conclusion
Calibration, verification, and validation are the 3 terms that most of us used interchangeably. In this post, I have presented the difference of each term, the reason why we need to perform them, simple examples and the necessary requirements under the clauses of ISO 9001 and 17025 standard that requires the use of calibration, verification, and validation.
By having the knowledge and understanding regarding the meaning of each term, we can easily understand and interpret what the standards are requiring us to do.
Still confused and unclear between the differences in calibration, verification, and validation? Please feel free to comment.
You may also visit the specific example where these 3 terms are used in a Liquid Oxygen Tank System in this LINK.
For other important terms, check my other post: Differences Between Accuracy, Error, Tolerance, and Uncertainty in a Calibration Results
Do not forget to share and subscribe.
You can follow me on my FACEBOOK Page
Edwin
33 Responses
Alvin Laborte
can you give an example instrument with specifiaction that we can determine or differentiate calibration,verification and validation?
edsponce
Hi Mr. Alvin,
Thank you for reading my post.
I have created a new post to answer your concern.
Check it out in this link and tell me what you think.
http://bit.ly/example_CAL_VER_VAL
Thanks and regards,
Edwin
Adrisman Yunus
Hi Edwin,
I was not able to access the example link. Can you let me know how can I access it…?
Thanks
Idris
edsponce
Hi Adrisman,
Thanks for informing me.
Please visit this link>> https://calibrationawareness.com/how-to-differentiate-calibration-verification-and-validation
Thank you for visiting my site.
Best Regards,
Edwin
edsponce
Hi Adrisman,
Thanks for informing me.
Please visit this link>> https://calibrationawareness.com/how-to-differentiate-calibration-verification-and-validation
Thank you for visiting my site.
Best Regards,
Edwin
Lance
Great discussion and provides a good understanding of often misunderstood terms. I’m currently using your Calibration or No Calibration Criteria Cheatsheet. As a recommendation during this assessment, it may be of value to incorporate when a verification, validation or calibration is the most appropriate for the intended M&TE.
edsponce
Hi Lance,
I am happy to know that you are using the Calibration or No Calibration Criteria Cheatsheet. I hope that it served you with your purpose.
Thank you for your recommendation. This opens up a new understanding of using the Cheatsheet.
Since we have learned the difference between calibration, validation, and verification, I also recommend that the cheatsheet will be used where the applicability of the M&TE (Measuring and Test Equipment) falls under these three terms (processes).
So the importance of the cheatsheet will be applicable during the assessment of all M&TE used for calibration, verification, and validation.
Therefore, our knowledge in the assessment is now better, that once an M&TE is used during calibration, verification, and validation, it will be wiser (or obvious) to include it within our assessment if it needs or does not need a calibration service.
Below are some additional thoughts:
– If an M&TE is used to calibrate another M&TE, then it needs calibration;
– If an M&TE is used to verify a product to determine either pass or fail based on a given tolerance, then it needs calibration.
– If an M&TE is used or part of a validation process to check product safety, reliability, and quality, then it needs calibration.
Best Regards
Edwin
Ariel Glenn
Thanks Edwin for this informative topics , still there are many people don’t know about calibrations, some of them say it just comparing two instruments, like what you said upon your topics about the difference between validation and verification and uncertainty, they didn’t know the real meaning each of those words, your topics are very helpful and informative, more power to you my friend
edsponce
You are very welcome my friend. Appreciate your comments.
Best Regards,
Edwin
kainat
what is difference between calibration and monitoring?
edsponce
Hi Kainat,
Appreciate the time you spent reading my post.
Calibration is what we perform for a specific measuring instrument to detect its accuracy or inaccuracy in comparison to a reference standard (a master). In addition, since we can determine the error based on the calibration result, we can also verify if it is within its accuracy or tolerance.
Monitoring is an activity or a process that we perform within a specified time frame in order to analyze, evaluate, verify conformity to requirements and come up with a solution either to correct or improve a certain process based on the monitoring results. During monitoring, we use a measuring instrument to gather data, and in order to have an accurate monitoring result, the measuring instruments must be calibrated.
Because of your question, I was motivated to write a separate post where I explained more in detail about monitoring, measurement, and calibration as applied to the calibration process as per ISO 9001 and ISO 17025.
Check this link to visit the post.
Best Regards,
Edwin
Zach
Hello Edwin,
I am implementing ISO 17025 in our lab. These posts you have made have answered several of my questions. I have a question regarding calibration following your explanation above.
I am trying to distinguish between indirect verification and in-house calibration for our hardness tester. I understand the ASTM method verification.
If I am verifying my hardness tester with reference standard hardness blocks, what would I use to calibrate my hardness tester? Would calibration be performed using the hardness blocks to determine a correction factor for the tester, and verification then checking that test results after correction is within verification error%?
Any suggestion would be greatly appreciated,
edsponce
Hi Zach,
You are correct in calibrating your hardness tester with a standard block since the standard block is your reference standard as you mention.
Depending on the model of hardness tester, other standards that can be used to calibrate it is a load cell or standard weights.
Since you have a standard block as the reference standard, this is the one you will use for calibration and verification. Make sure that your standard block is calibrated by a qualified lab.
Below is my suggestion, and as I see, it is aligned with your question.
As per your in-house calibration procedure, you will gather data where you compare the hardness tester reading with the standard block nominal value.
With these results, you can calculate the error and the correction factor.
And in order to determine if your results are acceptable or not, a pass or a fail, this is where verification comes in.
With the same data, check the readings if it is within the tolerance of the hardness tester, or with your process tolerance.
If the results are acceptable, it is up to you if you will perform an adjustment or not. But if the hardness tester has the capability to be adjusted (I believe it has), better to perform it, otherwise, use the correction factor to compensate for the error.
I hope this helps.
I appreciate your time reading my post.
Edwin
Charlie
Sir, does this mean that all the instruments that were calibrated must also be validated? Who performs the validation? Thanks
edsponce
Hi Charlie,
Actually it is a NO, it depends on the situation on how you will use that instrument. But all instruments that are calibrated should be verified.
Below are some instances or situation where we need to perform validation:
1. If the instrument is newly installed,
2. The instrument will be used for the first time
3. If you change the setup or transferring the instrument to other locations
4. Specifications or product design has been changed
Before you release or approve your measuring instruments to be used for your product, then you need to perform validation in order to check its suitability for your intended purpose.
In the calibration process, what is being validated is the method used for calibration, which includes the instruments and reference standard. The method is also known as the calibration procedure, either standard method or non-standard method.
In order to use the required method, validation is first performed to determine if the method you will be using with respect to the reference standard you have is suitable to be used.
Based in ISO 17025:2017, clause 7.2.2.1, below are some techniques that you can do to perform validation, it can be one of or a combination of the following:
1. Evaluation of bias and precision
2. systematic assessment of the factors influencing the result;
3. comparison of results achieved with other validated methods;
4. interlaboratory comparisons;
5. evaluation of measurement uncertainty
During validation, you should have validation procedure and a validation record where all the necessary results and a statement on the validity of the method, detailing its fitness for the intended use is stated.
Validation should be performed by the person who has enough knowledge and expertise about the instruments supported by the person who will approve the validation performed. These details should be reflected in a validation report.
I hope this helps,
Edwin
Joe Monahan
Q: What are the limits of in-house calibration?
edsponce
H Joe.
Thank you for visiting my site.
Please give more details about your question, in terms of what limits are you refering to?
Thanks and regards,
Edwin
Nico
Hi Edwin! VIM, ISO, multinationals, etc, and your page is still the most clear. Thanks for your contribution.
I think I have the same doubt as Joe, but let me try to clarify. In your post, you state that the differences between verify and calibrate are:
a. Traceability
b. Computed uncertainty
That means that both verification and calibration process involves:
a. Measuring n times the UUC (if it is a reference material)/measuring n times WITH the UUC (if it is a measurment equipment)
b. Comparing those measurements with a standard (that can be either a calibrated reference material or another calibrated measurement equipment)
c. Computing the correction factor
And then deciding if recalibrate, adjust, extend usability, scrap or make obsolete.
If that is true, then we as an ISO 17025 accredited lab:
1. Perform periodic external calibrations for essential measurement equipments involved in accredited essays.
2. Perform periodic verifications/¿or in-house calibrations? with adjusments for non-essential measurement equipments, using the external calibrated equipment as standard.
And the final quesion is:
– Why the in-house calibration process breaks the calibration chain traceability? Does it needs to be validated by someone in particular, in order to be considered as part of the chian?
– If we perform a uncertainty computation as a part of the in-house calibration process, would that difference be cancelled?
Hope I didn’t get to fuzzy. thanks in advace for your answer!!
edsponce
Hi Nico,
You are welcome, I appreciate your feedback.
Regarding your concern, Yes, Calibration involves traceability and measurement uncertainty. When these 2 are present, then the calibration performed is acceptable. During verification, we will be using the result from calibration (the ‘error’ result), for us to determine if the UUC has an acceptable performance based on the tolerance limit.
As we can see, ‘verification process’ needs to meet the requirements of ‘calibration process’ in order to have a valid decision in verifying a result.
I do not see that in-house calibration breaks the traceability chain. In order for a calibration process to be traceable below are some of the evidences that we need to have or provide:
1. Using a standard calibrated by higher lab using a traceable standard,
2. With measurement uncertainty
3. Calibrated using a recognize method
4. With accreditation logo in the calibration certificate.
5. And more…
Measurement uncertainty alone does not ensure proper traceability, but it is one of the main requirements and evidence of traceability because this is what we actually pass on to the lower end of the traceability chain. If we have at least two of the above list, I believe our in-house calibration process has an acceptable traceability. You may want to check my separate post about traceability in this link >> Traceability
Thank you for the time reading my post and commenting as well.
Have a nice day.
Edwin
sean calvelli
Hello,
What specific equipment do I need to self- perform verification on the following Hand held tools:
Digital multi meters, torque drivers, wrenches, and calipers
Thank you
edsponce
Hi Sean,
Your concern will fall under the process “intermediate check” and therefore I recommend below verification technique:
For multimeter – if you do not have a multi-product calibrator to source out an electrical signal (like Fluke 5522a), you can use another calibrated multimeter. Measure any electrical output signal that is close to the range you are using. Just compare their readings.
For calipers, a gauge block is the best standard for verification, but if you do not have one, then you can use a previously measured item or product with recorded results.
For torque drivers or wrenches, the principle above can be applied if it has a digital display that you can read and compare with. But if it is purely mechanical, it is difficult to perform a verification if you do not have a calibrator, therefore, only a physical and functionality check is performed.
If you observed any abnormalities, like a broken seal or a damaged part during checking, then you need to have it recalibrated.
This is I believe part of your quality check to maintain the confidence in your measuring instruments.
I hope this helps, thank you for reading my post.
Edwin
Mohamed habashy
difference between tolerance and uncertainty
edsponce
Hi Mohamed,
Tolerance
It is the maximum error or deviation that is allowed or acceptable as per the design of the user for its manufactured product or components.
If we perform a measurement, tolerance value will tell us if the measurement we have is acceptable or not.
If you know the tolerance, it will help you answer the questions like:
1. How do you know that your measurement result is within the acceptable range?
2. Is the final product specification pass or fail?
3. Do we need to perform adjustments?
“The bigger the tolerance, the more product or measurement results will pass or accepted.”
For example, A pressure switch is set to turn on at 10 psi. The process tolerance limit is 1 psi. Therefore, the acceptable range for the switch to turn on is in the range of 9 to 11 psi, beyond this range, we need to perform calibration and adjustment.
Uncertainty
Also known as measurement uncertainty. It is always defined as the quantification of the doubt. If you measure something, there is always an error included in your final result (a doubt – no result is perfect) since there is no exact measurement results.
Since there are no exact measurement results, what we can do is to determine the range where the true value is located by calculating the measurement uncertainty.
We do not know the true value is, what we know is that the true value lies within the range that we calculated, the measurement uncertainty result.
“The smaller the measurement uncertainty, the more accurate or exact our measurement results.”
For example, a pressure switch will turn on at a setting of 10 psi with a calculated measurement uncertainty of +/- 0.3 psi. This means that the true value of the setpoint is located in the range of 9.7 to 10.3 psi.
To learn more, visit my other post in this link >> Calibration terms
Thanks and regards
Edwin
Mohamed habashy
Appreciate your response, could you tell me about the relationship between tolerance and reproducibility regarding astm.
edsponce
Hi Mr. Mohammed,
I do not know what ASTM are you referring to but below are the relationship of Tolerance and reproducibility:
Tolerance is the maximum error or deviation that is allowed or accepted in the design of the user for its manufactured product or components.
Reproducibility is a result of repeated measurements by, let say, a different person, repeating the same measurement (example: 10 measurements performed by a different person).
For simplicity, the results of the reproducibility may be the average of all the results or the standard deviation that is being compared to a tolerance.
I hope this helps,
Edwin
Arnel Ruiz
Hi Edwin,
On the external laboratories who provide calibration services with Certificate is valid until 2022, with the ISO 17025:2005 accreditation certificate and operation of laboratory quality management system that meets the principles of ISO 9001:2008 instead of ISO 9001: 2015 & 17025:2017 version they can able to carried those accreditation without reassessment for the new version
regards
Arnel
edsponce
Hi Arnel,
I am not sure about ISO 9001 but with ISO 17025, it has a 3 year transition period where it is still valid until this year, 2020. After this transition period, all labs should be accredited to the 2017 version. Maybe they have already a plan for the transition, you need to ask for it as part of your supplier evaluation.
Thanks for visiting my site.
Best Regards,
Edwin
M. Saleh
Hi Eng. Edwin,
As we do in every calibration we verify of accuracy of manufacture & the tolerance of end user, is this enough or instead of what you talk about?
Thanks in advance.
edsponce
Hello M. Saleh,
It depends on your objectives or activity involve.
If you are just performing calibration for a customer’s instruments, then what you have stated is enough. But when your objective is to create or validate a calibration procedure/method, then you need to perform what I have just presented.
Thanks again for reading my post.
Edwin
Hossam Badr
Hi Eng. Edwin
regarding the Digital Multi-meter Calibration,Is there a standard that controls the interval between each calibration process?
thanks in advance
edsponce
Hi Eng. Hossam,
Good Day!
There are no specific standard that controls the calibration interval or frequency of calibration for a Digital Multimeter even for other instruments. The control of the Calibration interval depends on the user. Some users follow the manufacturer’s recommendation.
But below are some that dictate the calibration interval:
1. Based on the performance history of the instruments (like stability);
2. Based on the frequency of use;
3. Based on the regulatory bodies (example: required by the government);
4. based on the experience of user with the same kind of instruments.
5 and others…
There are available guidelines that we can use to guide us on how to determine the calibration interval, you may check out this standard document: OIML D 10 :2007 -Guidelines for the determination of calibration intervals of measuring instruments.
I hope this helps,
Edwin
Abu
Hi
Appreciate if you could share some of your thoughts on the following scenarios
1) calibrated thermometer verification (-50 to 200oC) with tolerance range of +- 1oC throughout the calibration range.
Routine use : 2-8, -20, 60
Since the tolerance is actually the same the whole calibration process the lab would like to propose only 1 point verification against a reference thermometer for the whole of the calibration range.
2)variable volume micropipette is calibrated using ISO8655 requirement at nominal, 50% and 10% volume. The guide states that the MPE for selected variable volume will follow the MPE set for the nominal volume. the lab would like to propose either
a) only 1 point verification for the whole of the calibration range.
b) if verifcation conducted at the same calibration point; if either one fail equipment will be considered to be out of tolerance
thank you
Abu
edsponce
Hi Abu,
For the Fist question — I recommend to perform 2 points, 1 for positive range and 1 for negative range. Most temperature sensors have different specification on a negative range compare to positive ranges. Check the accuracy rating in the manufacturer specifications to be sure.
For the second question — I suggest to perform 1 point verification to the range which you are always using. The closer the better.
As a suggestion for the whole process, applicable to both questions, verify first the whole range, better 3 or more times. Then if you observe that during this verification it has passed your expectations, then it is time to push through on what you have shared with me.
You will then also have a data that can be used as an evidence in case an auditor will question you.
I hope this helps.
Thanks for visiting my site.
Edwin