As a Calibration Technician, an Equipment Engineer, a QA, or just a user of a calibration certificate, let me ask you below questions:
Q1: Do you know how to interpret a calibration certificate during measurements and/or preventive maintenance?
Q2: If you receive a newly delivered calibration certificate, what would you do?
Q3: What are the important parts of a calibration certificate we need to fully understand?
Q4: What are the requirements of a calibration certificate based on ISO 17025?
Q5:How can you determine and understand if the instruments you are using are performing as expected?
In this post, I will share with you the importance of a calibration certificate review, discuss its true purpose, and how to use it properly. I will also explain and answer the questions I listed above.
Once you understand its features (I will call it features because of its application), you will appreciate the importance of a calibration certificate while performing measurements, preparing for an audit, or just simply evaluating the performance of your measuring instruments.
I will also share the requirements of a calibration certificate based on ISO 17025 as applied to an accredited calibration laboratory that every Calibration Technician must know.
What is a Calibration Certificate?
Imagine you have a pressure gauge, and you want to make sure it measures pressure readings correctly. A calibration certificate is like a report card for instruments that are used for measurement, like thermometers, scales, or rulers. It tells you if these instruments are working properly and measuring things accurately according to requirements.
A calibration certificate is a record of the results of calibration performed on a specific Instrument or measuring equipment. This is also known as a ‘certificate of calibration’ or simply a ‘calibration report’.
Some of the Important Uses of a Calibration Certificate are the following:
- Verification of Accuracy
- Evidence of Compliance with Standards
- Metrological Traceability
- Quality Assurance
- Decision-Making
- Risk Mitigation
- Customer Confidence
- Equipment Maintenance
- Audits and Accreditation
- Legal and Regulatory Compliance
What is an ISO 17025 Accredited Calibration Certificate?
I believe most instrument users are aware of what a calibration certificate is. However, I am not sure if all who use a measuring instrument with a calibration certificate know how to interpret calibration certificate content.
An accredited calibration certificate is a certificate that follows a specific format guided by the accreditation body, and contains the following information:
- Issued by an accredited calibration laboratory
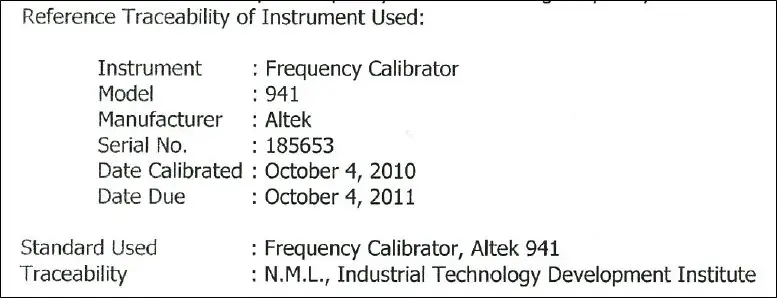
- It has traceability information, these are:
- The details of the reference standard used
- The measurement results
- The estimated measurement uncertainty results
- The name of the Higher Lab or Organization where traceability originates (ex: NIST)
- It has all the elements that I listed below. It complies with the requirements of ISO 17025:2017 under clause 7.8, reporting the results.
- And of course, it has the logo of the accreditation body.
To ensure that the lab is accredited under ISO 17025, we should have a copy of their accreditation certificate and scope as evidence that they are accredited.
This is to differentiate it from other non-accredited calibration records where there is no measurement uncertainty result and with limited information.
These kinds of certificates are just a verification result/record.
Mostly, what I observe in most users (quality personnel, safety officers, or other technicians) is that they only use the calibration certificate as evidence that the measuring instruments that they are using are calibrated.
Only a show part for the auditors telling ..“Hey Mr. Auditor, our Instruments are calibrated, we have here the calibration certificate, we have complied with your requirements”.
And some technicians, only use the calibration certificate to verify the date and due dates if the instruments are still within the said calibration date and nothing follows.
Sometimes, calibration certificates are kept directly without reviewing them.
We need to change this habit and start using the calibration certificate for its intended purpose.
Key Elements of a Calibration Certificate Based on ISO 17025:
- A title (calibration certificate).
- The name and the Address of the Calibration Laboratory.
- Unique identification number of the calibration certificate and the pages until the last page of the calibration certificate ( an example is a serial number; page 2 of 2).
- The name and contact details of the customer.
- Identification of the calibration method.

Other evidence of traceability that can be seen in the calibration certificate are:
- The LOGO of the accreditation body
- Measurement Uncertainty results
- The details of the reference standard used
- The Measurement results

>> It means that the calibration certificate cannot be reproduced (to make another copy) in part only where some page is missing (in cases where there are multiple pages). Certificates are allowed to be reproduced considering that it was reproduced with a complete number of pages. This will ensure that the user will get the complete information, and therefore, proper interpretation of data results. In any case, if you want to reproduce it with one part only (1 page only), ask first for the approval of the laboratory.
>> To understand easily, let us take the opposite statement, which can be: “The calibration certificate can only be reproduced in complete pages only.”

We should consider the above list when creating a format for a calibration certificate. Following the requirements above will ensure that all relevant details for a calibration certificate will be covered.
Their location in the calibration certificate is not the same, some certificates are one-paged only while others are 2 pages or more. It depends on the measuring instruments being calibrated.
Furthermore, as per Clause 7.8.1.3, states that “When agreed with the customer, the results may be reported in a simplified way”.
This means that some Information stated above can be omitted or not be reflected in the calibration certificate as long as it is agreed with the customers or users. But make sure that the omitted information is available in the records or raw data. F
What To Do Once You Have Received a Calibration Certificate?
We must review the calibration certificate upon receipt. But what needs to be reviewed, do you know how to review a calibration certificate? We can use the above list as a checklist when reviewing a calibration certificate or verifying the completeness of a calibration certificate.
If you received a calibration certificate from a calibration laboratory, it should contain the above list or ensure that it follows the above requirements. More specifically when the calibration laboratory that we have outsourced is not accredited to ISO 17025.
If you are under quality or handling calibration, one of your main concerns is to check all the above requirements of a calibration certificate to verify if complete or properly labeled and spelled.
One mistake for example, on a serial number, will invalidate the traceability of the instrument, traceability in the sense that the identification of the specific instrument is lost.
In addition to most details, I want to emphasize the list of requirements above which is the “Statement of compliance with requirements and specifications”.
This should be reviewed or checked immediately in the certificate to ensure that newly calibrated instruments have passed the calibration and are safe or acceptable to use. Below is a sample statement.

There are some cases where you will encounter an “Out of Tolerance” result upon receipt from the external calibration lab.
Determining this in the first place will prompt you to apply the corrective actions. (please see below info on how to check the results or data under as found/as left)
If ever you encounter an OOT (Out-of Tolerance) result, one corrective action is to adjust or reduce the calibration interval. How to perform this? Check out my other post here>> calibration interval adjustment
What to Inspect in a Calibration Certificate During Measurement, Intermediate Check, and/or Preventive Maintenance?
Many of the technicians performing measurements, intermediate checks, and/or preventive maintenance do not check or review the calibration certificate concerning the result of calibrations or the measured output values of their instrument.
They are already confident that the calibrated instruments they use are accurate. This is what I usually hear, ”My instrument is accurate because it is newly calibrated!”.
Yes, they are correct at some point, but not always…if this is how you think about calibration, then you are missing something. Not all calibrated instruments are being adjusted during calibration and therefore, it is not as accurate as you believe.
Once a newly calibrated Instrument is received with a calibration certificate, of course, one must know how to review and interpret the content of the calibration certificate.
This is not just a job of quality personnel (calibration technician) but specifically, the one who directly uses the instrument, the technician, or the operator.
But if you are a technician or an engineer directly using the instruments, in addition to the above requirements, concentrate on reviewing the following checkpoints:
- As found /as left data
- Traceability information
- Uncertainty results
- Correction/error
Usually, the above contents can be seen on the second page of the calibration certificate, which we commonly call it calibration results (for the traceability details, mostly it is on the first page).
How to Use the Calibration Certificate during the Measurement Process and/or Preventive Maintenance?
We need to determine and understand the calibration certificate on this part as an engineer or technicians simply because it has a significant effect on our measurement process. Most of the calibration results have what we call a Correction Factor.
To understand better, below is an example:
Correction or Error
What is a Correction Factor and how to calculate a Correction Factor in calibration results?
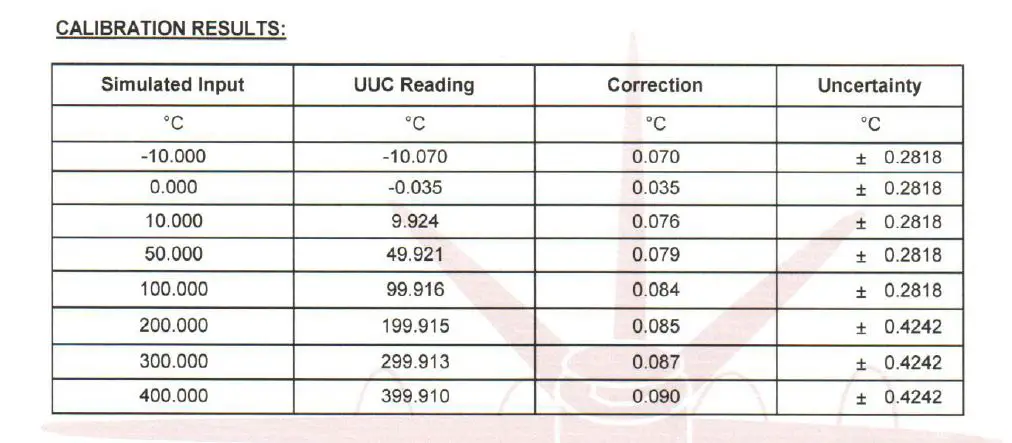
When we are measuring temperature, for example at 0 settings using our newly calibrated thermometer, we notice that at zero points; it has a correction of + 0.035 (where the error is -0.035).
Therefore, every time that we make a zero-point measurement, our actual value should be added with a +0.035. The same thing with the other test points.
We call this value a Correction Factor (CF). It is a value that we either add or subtract in the actual measurement to compensate for the error. The ‘Correction Factor’ is the opposite of Error.
It is simply the difference between the STD value and the UUC results. To calculate the correction factor, just subtract the ‘UUC reading’ from the ‘Nominal Value’ (STD-UUC).
Below is an example based on the table above.
Example:
…… Nominal Value = 10.00
…… UUC reading = 9.924
…… Therefore: 10.00 – 9.924 = 0.076, which means that the correction is +0.076
.
This is important if we are targeting a smaller error or implementing a strict tolerance. In this way, we will ensure that the results we get are the most accurate ones.
Most technicians do not use this correction factor because they do not review the certificate they received or do not know its purpose (luckily, they have a wider tolerance in which correction does not matter).
They simply use the instruments and know that it is already perfect because it is calibrated.
The correction factor is very important in Measuring Instruments or machines that can be adjusted physically or electronically.
An example of this is during preventive maintenance, once we determine the correction factor, we can input this on the calibration part or program of the equipment and compensate or correct the errors.
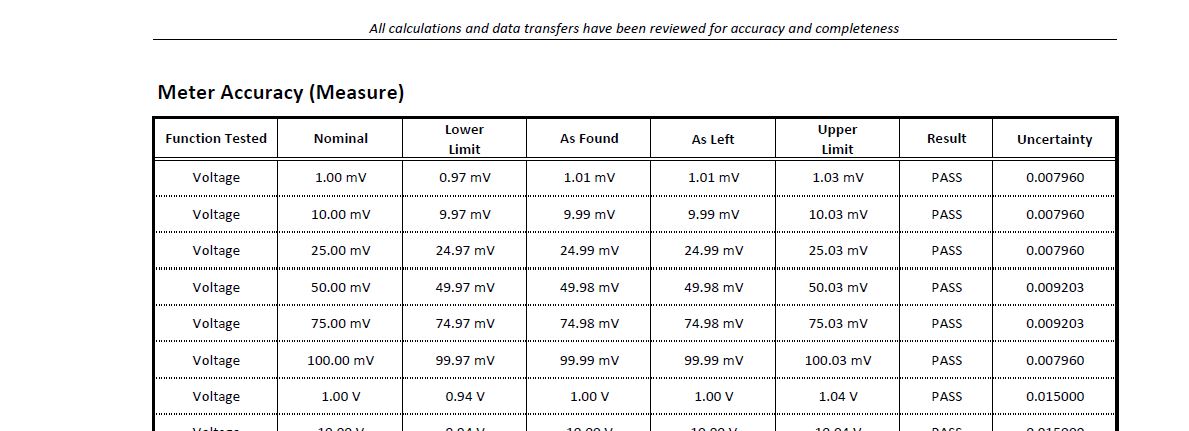
As found/As Left
If you observe the figure above, it has the ‘as found’ and ‘as left’ columns. This is the “before and after” we calibrated the instrument.
It has no correction but as long as the measurement value lies within the limit, your measurement is acceptable. The ”As Left” column will be different just in case an adjustment was done if ever the “As Found” value is outside the limit.
As an engineer or quality personnel, one should check if ever the “as found” is within the upper and lower limit. Just in case it is out of this limit, it means that your instrument is “out of tolerance” before it was calibrated.
By being aware of this information, you can make a proper adjustment and perform corrective action and assessment to scope up the affected units or parts where these instruments are used.
To understand more regarding data interpretation, visit my other post >> Differences Between Accuracy, Error, Tolerance, and Uncertainty in a Calibration Results
Read more about Correction Factor in this link >> Simple Ways to Apply Correction Factor in a Calibration Certificate If the Correction Factor you Need is not Given.
Uncertainty Results and Traceability
One of the ways to determine that we have a traceable calibration is to check that the calibration results have a computed or estimated measurement uncertainty result.
This will ensure traceability because, during estimation or computation, the accredited laboratory’s BMC (Best Measurement Capability) is included, which they also acquire from a higher laboratory standard.
What is measurement uncertainty? Measurement uncertainty is a computed range of value, usually an estimate to quantify the doubt that exists in a certain measurement result of a specific instrument.
It is the combined effect of the most valid sources of error with a confidence level of 95%.
For example, on the measured voltage above, 1 mV is the true value where the measured or actual value is 1.01 and the uncertainty result is 0.007960 or (1.01+/-0.007960)mV at a confidence level of 95%
This means that we are 95% sure that the measured value lies within the limits of 1.00204 to 1.01796 (+/-0.007960) mV. The smaller the uncertainty, the better the accuracy.
With this result or statement, clearly, uncertainty and error are NOT the same.
Where to use these uncertainty results?
In creating a specification to determine the tolerance of a certain instrument, we can use the uncertainty results as a basis, besides the Manufacturer’s specification.
Uncertainty results are also used during the assessment of compliance to specifications, including tolerance, to determine pass or fail results.
If we are also performing calibration and estimating the uncertainty of measurements (this is another topic), we will use the uncertainty results as part of our uncertainty budget, a type-B source of uncertainty computation.
To see the presentation regarding the use of actual uncertainty results, visit my other post
>> Differences Between Accuracy, Error, Tolerance, and Uncertainty in Calibration Results
>> 8 Ways How You Can Use the Measurement Uncertainty Reported in a Calibration Certificate
How Do I Know that the Calibration Certificate I Issued Meets the Requirements of ISO/IEC 17025:2017 Accurately?
If you are a calibration lab issuing a calibration certificate, there are 2 ways you can check the content of the calibration certificate:
- By comparing it to the listed calibration certificate requirements specified under ISO 17025:2017 clause 7.8.2.
To determine if you covered all the requirements of a calibration certificate, you can use ISO 17025 clause 7.8.2 as a checklist. All the requirements for a calibration report are listed there.
One way to ensure that all requirements are followed consistently is to create a calibration certificate template. - By verifying the requirements of the customer.
Customer requirement is one of the main checkpoints when reviewing a calibration certificate; therefore, we must ensure that it is accurately followed.
It is mostly seen in the calibration results. The customer requirements are taken during contract review where the lab and the customer have an agreement before the work is accepted.
This should be documented for example, on the Quotation or purchase order (PO), sometimes, there is a customer request form. - By following the Requirements of the Accreditation Bodies
Another requirement that we need to follow and implement when it comes to the completeness of our calibration report is the requirements of the accreditation bodies.
There are certain requirements that we need to follow, where failure to implement these may result in non-conformance, and worse, revocation of accreditation, some of these requirements are:- Use the exact type or format of the accreditation body’s logo
- Correct placement of the accreditation body’s logo in the certificate
- Using their logo and accreditation number when issuing a calibration report with parameters not covered in the accreditation scope.
Before releasing or before the approval of the calibration certificates, these should be checked to ensure the correctness and accuracy of the calibration report content.
Quick Summary (TL;DR)
- A calibration certificate is a measurement tool, not just a document
- Always check error, correction, and uncertainty
- Apply correction factors when needed
- Use uncertainty in decision-making
- Verify traceability and compliance
Final Takeaway
A calibration certificate only becomes valuable when you use it.
If you only read it, you gain information.
If you apply it, you gain accuracy, confidence, and compliance.
❓Frequently Asked Questions (FAQ)
1. What is the purpose of an ISO 17025 calibration certificate?
It provides verified measurement results, uncertainty, and traceability to ensure accuracy and compliance.
2. How do you interpret calibration results?
Compare measured error with tolerance and consider uncertainty before making decisions.
3. What is the difference between error and correction factor?
Error is deviation; correction factor is the value applied to adjust readings.
4. Why is measurement uncertainty important?
It defines the confidence range of your measurement results.
5. Can I use a calibration certificate without applying correction?
Yes—but it increases measurement risk and reduces accuracy.
Struggling with Calibration Certificate Interpretation? Check out my ebook at this link >> DEMYSTIFYING THE ISO/IEC 17025 CALIBRATION CERTIFICATE
Conclusion
We have determined what is a calibration certificate and its importance in our industry and in our measurement process. The need to review a calibration certificate and what are the necessary checkpoints which are based on ISO 17025 requirements are discussed. And finally, the important requirements to consider for technical personnel in the calibration certificate when measuring or performing equipment preventive maintenance are also presented.
In using a calibration certificate, one must also be familiar with the principles of calibration through training on calibration awareness.
Thank you for visiting my site, please leave a comment, subscribe and share.
Best Regards,
Edwin




79 Responses
Jerry
Thank you for this very useful information, additional for ny knowledge.
edsponce
Hi Jerry,
Your Welcome. I appreciate you liked and learned something from it.
Best regards,
edsponce
Rakesh
I learned a lot from all your posts. Thanks for sharing your knowledge with us. And also please post calibration management of ISO 9001
edsponce
Hi Rakesh,
Your Welcome and thank you for taking time reading my post. appreciate that you learned from it. I will consider your suggestion about Calibration Management for ISO 9001.
For the moment, if you did not read yet my related post about In-house calibration, here is the link..https://calibrationawareness.com/elements-in-implementing-internal-calibration-laboratory
Best Regards,
Edwin
Rakesh
Thanks for your reply Edwin, i read the post about in-house calibration, but i would like to know about external calibration laboratory management. And you mentioned the temp & humidity limits to be maintain in in-house calibration. Am staying at sea areas so generally the temp and humidity is very high over here, like 32 deg and 75%rh in normal days. but at the same time as you said we have to consider the equipment specifications. so if the temp & humidity values at laboratory are within the limits of specifications on the equipment, can i carry out the calibration at 32 deg and 75%rh?? and i will mention these environmental conditions on the calibration certificate so if auditor ask me about that,Then, if i tell him the same reason like those are within the limits of operational conditions suggested by manufacturer, will the auditor agree for it or not?? please clear my doubt. because as an external vendor most of the instruments to be calibrated at on site… so it is very difficult to control the environmental conditions… hope u will understand my intention.
edsponce
Hi Rakesh,
Humidity and Temperature is not that big deal for an Electrical or pressure measurements because they have a wide acceptance operating range unless it is in a dimensional calibration where you are using a gauge block and change in temperature is very critical.
I understand your intentions and I agree to what you have said. Your concern is how can you answer just in case the auditor will ask you regarding the environmental conditions outside the laboratory which is different inside the lab.
The specified humidity and temperature is applicable only inside the lab because we can control it and it is required by an international standard. But during on-site calibration, we cannot control it so the best thing to do is to control or prepare some quality control procedure before performing field calibrations. The best question now is, how to we prove that we have an acceptable calibration even in a different environmental condition?
In your first question, Yes, you can still carry out and record the calibrations in that environmental conditions, we are also doing that. Most of the instruments are best calibrated on the site or location where they are being used. The highest temperature I experience while performing a calibration is 46 Deg C.
When the auditor ask you, just prove to the auditor that your standard is capable to that environmental conditions by having it documented in your procedure basing it on its specs, I believe you have a procedure for internal calibration, then you may add this part.
The most important things to consider like I said is to perform a quality control procedure, we call it an Intermediate Check or Functional check. Before you let your standard out of the Lab or office, perform a functional check,.Just perform a simple calibration check by measuring a certain value. Then when the standard will return back to the lab, perform again the same measurement. Record everything. This is to ensure that the standard or equipment used is still in tolerance even exposed to different temperatures and other factor like vibrations while performing on-site calibration.
Based on my experience, this is the only thing you need, just prepare all these documents and all will be fine just in case an auditor will ask or investigate. And lastly, include in your report or record the site calibration performed is a request or approved by your customer.
I hope this helps.
Edwin
Rakesh
Thanks a lot edwin. You are a great teacher.
edsponce
You’re welcome, happy to help.
best regards,
Edwin
Antony
Hi
Thanks for your valuable article it is worth reading
Can you send me how to calculate uncertainty for pressure gauge calibration report
Also Temperature instrument and other process instruments
Antony, antonypallan@gmail.com
edsponce
Hi Antony,
I am glad you liked my article. What do you mean about uncertainty calculation, is it how to use the uncertainty result indicated in the calibration certificate to be used in uncertainty computation or how to calculate uncertainty of instruments to be displayed in a calibration Certificate?
Best Regards,
Edwin
Antony
Dear Edwin
How to calculate uncertainty of instruments to be displayed in a calibration certificate
Thanks in advance
Antony
edsponce
Hi Antony,
Do you have some background about uncertainty computation? for me to know how much I can help you about the subject.
Regards,
Edwin
Antony
I have gone through the papers available in the net.
I want a excel sheet for measured value entries and master instrument details . The uncertainty result should be displayed in the calibration report. Our present report does not calculate the uncertainty component
We are doing calibration of Pressure & Temperature gauges, Digital Temperature indicators, Pressure & Temperature Transmitters etc used in Chemical process industries
Regards
Antony
BachNga Le
Hello
If the certification show 2 points at 0 and 50 C, how could I figure out the correction factor at 32 C?
edsponce
Hi Bachnga,
Thank you for reading my post.
There are 3 ways you can do to determine the best correction factor, these are:
1. You can choose the one that is nearer to the value of your test point. In your example, 32 is nearer to 50, so use the correction factor (CF) for 50.
2. You can perform averaging of the 2 nearest range. For example, 0 has a CF of 1 and 50 has a CF of 3, add 1 and 3 then get the average (1+3 = 4; 4/2 = 2).
3. If you have so many data results where the correction factor is increasing with range, use the linear interpolation (many examples if you google ‘linear interpolation’).
I hope this helps.
Best regards,
Edwin
Rahman
Hi sir,
I just read your answer as this issue is also my recent concern. A great article for my reference.
I just want to ask some questions if u don’t mind.
1) Referring to your answer no. 2, after averaging the 2 nearest range and the answer is 2, at 32c we can use the correction factor of 2 instead of using the correction factor of 3?
2) May I know which guideline you referred to? Can you show me or provide me the guideline regarding this issue?
Thanks.
edsponce
Hello Rhaman,
You are correct. I am using the averaging method to determine the best estimate for evaluating results.
Regarding the reference, as per my understanding, there is no single answer for it, it also includes experience and professional judgment utilizing the results from statistical methods or techniques.
There are other more detailed references out there that are not in my awareness but I will share with you one of my references. You can use ISO 17025:2017 as a reference. Below are the requirements under clause 7.7 :
7.7.1 The laboratory shall have a procedure for monitoring the validity of results. The resulting data shall be recorded in such a way that trends are detectable and, where practicable, statistical techniques shall be applied to review the results.
There are many statistic books and references that you can find online regarding this topic.
2 of the statistical techniques that I usually use and presented as an example are the mean or averaging and the linear interpolation.
You can also check JCGM 100:2008, Evaluation of measurement data — Guide to the expression of uncertainty in measurement.
I hope this helps,
Thank you for reading my posts.
Edwin
Rahman
Thanks a lot for your explanation. Keep up your great sharing. It’s really helpful.
edsponce
H Rahman,
You are welcome. I appreciate your comments.
Thanks for reading my posts.
Best Regards,
Edwin
Antonio
Hi Ed,
How about, the test points detailed in the certificate were 0,10, … .100% of range. there was a correction on 0% test point and 10% test point, your application (user) is at 5% test point for example, how will you apply the correction factor? is interpolation applicable to all type of testing/calibration parameters?
edsponce
Hellow Mr. Antonio,
Yes, it is applicable to all type of testing or calibration. You can perform either interpolation or average (add the result of 0% and 10% then divide by 2).
Thank you for reading my post.
Edwin
Charlie
Sir, I just want to ask if this is correct. This is how we apply correction during verification. If our analyst will weigh a sample of 26 g. Based on cal cert. when weighing a std mass of 26 g there is a correction of +0.0022 with uncertainty of +_ 0.0008 g. What i told them is that, when they weigh std mass w/c is 26g during verification, if the reading is withiin 26.0002 (true value of 26g) +_ 0.0008 you have to follow the correction factor w/c is +0.0022 but if out of range you have to apply a new correction factor w/c is true value – reading of std mass during verification. Is this correct interpretation sir? Thanks
edsponce
Hi Charlie,
During verification, you should use a mass with a known value, either a sample or a standard mass. A standard mass is better.
Now, since the balance has a correction factor of 0.0022, every time you measure a 26 grams sample, always include (add) the correction in the final result.
Please see the below presentation if this answers your question.
Given: (example only)
• Std reading (true value) = 26.0002
• Correction Factor = +0.0022 g
• UUC reading (actual value of balance)= 25.9967 g
• Uncertainty = 0.0008g
• Tolerance limit = +/- 0.005g
During verification, your objective or purpose is to check whether the instrument reading is within the expected tolerance.
First, determine your tolerance (25.9952 to 26.0052)
Second, put the standard mass, the std mass value = 26.0002, this is also your expected value to be displayed in the balance.
Third, record the actual value displayed which is equal to 25.9967
Fourth, this is the time to add or subtract the correction factor. In this case, we will add, therefore final value = 25.9989
Finally, compare the final value is within the tolerance.
Other users that do not need a highly accurate result do not include the correction factor if it is a PASS, but as a good practice to follow, since you have a correction factor, always include it to give a more accurate result.
Remember that the Correction factor is the result during calibration which is used to offset the error and therefore should be subtracted (or added) on the final value of the balance (UUC) results. A correction factor is applied to all instruments that cannot be adjusted, that is why we use the correction factor manually.
As per the uncertainty results, this depends on your procedure on how you use it.
Below are some ways on how to use the uncertainty results:
1. If you are performing measurement uncertainty calculation, this will be included in your uncertainty budget. (not related to verification)
2. You can use directly as your tolerance for verification.
3. You can include it within the calculation of your process tolerance. For example, Reference Std tolerance + instrument tolerance + uncertainty results = total process tolerance
4. Or you can use it to determine a pass or failed results. During analysis, a passed result means that even if you include the uncertainty in the results, it should still stay within the tolerance limits (decision rule as per ISO 17025:2017). See below example (same as above):
UUC reading = 25.9967
Uncertainty results = 0.0008
UCC + correction factor + uncertainty results = 25.9967 + 0.0022 + 0.0008 = 25.9997
Tolerance interval = +/-0.005 (25.9952 to 26.0052)
After the addition of the uncertainty results and still within the +/-0.003 tolerance, therefore it is a PASS. Or if it is outside the limits, then it is a FAIL.
The important thing is that how you use the uncertainty result should be stated in your procedure.
I hope this helps.
Edwin
Charlie
Sir in addition, what if during verification your uuc reading (of same std mass) is not the same with the uuc reading in calibration certificate. We performed verification every start of shift. And that is 3 times a day for ten months, based on verification logbook there were times that our analyst used new correction by simply true value- reading during verification. Is this correct sir? Thanks.
edsponce
Hi Charlie,
Yes, it is correct, that is the purpose why we perform verification, to see if it is still within tolerance or has an acceptable accuracy before we use the balance. If there is a big error, then we need to adjust or use the correction factor we obtain.
During verification of the balance, we do not need to use its calibration certificate. What we need only is the calibration certificate of the standard mass to obtain the exact value or true value.
Every time we take a reading from the balance, you compare it in the true value of the reference standard mass and use the correction factor immediately. I believe this is what you already do.
You can disregard the calibration certificate of the balance if you always use the standard mass for verification as long as the standard mass has a valid calibration report, and do not forget to record everything.
If you are monitoring the drift of the balance, this is the time where you can use the results from its calibration certificate. You compare the before and after results. This will show you its stability in the long run.
This is also one way of increasing the calibration interval of the balance once you have shown that it is stable in a defined period of time and use.
Thanks again for the comments.
Best regards,
Edwin
jhon lee
Dear Edwin,
I always see your good suggestion regarding calibration issues i have a one question regarding calibration certificate that if one company ISO 17025 accredited there accreditation scope cover temperature only after accreditation they buy a new master tools for pressure than how the will issue the certificate they mention the accreditation logo on his certificate of pressure or no because how they will tell the people that we are accredited lab if no logo on certificate even not in scope but lab is ISO 17025 accreditation.
i saw some companies not cover the scope but they put the accreditation body logo on his certificate please clarify for this.
Jhon lee
edsponce
Dear John Lee,
During the accreditation process, the accreditation body will check our competency based on the scope that we applied for. And once we have passed their accreditation, we can now use their logo.
Every accreditation body has a policy on the use of the accreditation logo on every calibration certificate that we use. I suggest you check this policy with your accreditation body.
The main point is that we cannot use the logo on the scope that we are not accredited. One rule when using the logo is that you can use the logo on a non-accredited scope considering that you will put a remark somewhere in the calibration certificate stating that “this scope or range is not accredited” or a similar term.
But to be sure, as I said, check or download the accreditation body’s policy regarding this matter. I believe it is downloadable in their website.
Your accreditation maybe in question once this will be audited during a surveillance audit if not fixed.
I hope this helps.
Thank you for visiting my site.
Edwin
rohit roy
what is relative confidence limit sometimes given in calibration certificate of CRMs? how to calculate tolerances using a central value when only relative confidence limit is given?
edsponce
Hi Rohit,
I did not yet encounter a relative confidence limit for a CRM.
The confidence limit is the value of the upper and lower end of a confidence interval. What I usually see in CRM calibration report is the standard deviation. As per my understanding, 1 Standard deviation is the same as 1 confidence interval which can be used as a basis for tolerance.
If you want, you can share your certificate with me and I will try to take a look. My email is edwin@calibrationawareness.com.
Thank you for reading my post.
Best regards,
Edwin
P. Gooljar
Dear sir, i just joined the your website and got opportunity to read your comments and valuable information published. Thanks a lot. P. Gooljar Mauritius
edsponce
Hi Pravesh,
You are welcome. I hope you will see articles that would help you more.
Thanks for visiting my site.
Best regards,
Edwin
Wahiba REMILI
Thank you very much for that report about certificate of calibration contents, much appreciated.
I would ask you for the meaning of:
” opinion and interpretation mentioned in this certificate is outside the scope of work”
I found it in the calibration certificate at the end as a “Note”, i didn’t understand what does it mean!!?
for information the data type is “As left”
Many thanks in advance for your help and clarification
edsponce
Hi Wahiba,
Opinions and Interpretations are part of the requirements of a calibration certificate/report. This part of the report is provided by the calibration lab.
It is only provided when appropriate or needed in order to give more meaning about the results that are reported.
Opinions and interpretations expressed are based on the calibration results as per the performed calibration procedure. These opinions and interpretations are given by an authorized person of the lab as per his expert assessment or understanding of the results.
Some instances where opinions and interpretations are required:
1. If requested by the user or customer;
2. If a result needs more clarifications regarding the use of the reported results;
3. Some limit of usage;
4. Special instructions on how to properly use the results;
5. Or any important interpretations that can help the user in using the reported results.
But in order for a person to give opinions and interpretations, he/she must be authorized by the lab.
As per the remarks,” opinion and interpretation mentioned in this certificate is outside the scope of work”, the ‘Scope of work’ means that it is what they are allowed to perform calibration or test as per the approved capabilities based on their accredited scope.
Accredited scope (also known as Scope of Accreditation) can be found on the second page of the accreditation certificate provided by the accreditation bodies under ISO 17025 Standards.
Since I did not see the calibration certificate, below are my inputs about the reason why the lab has indicated the remarks:
1. The result (or part of the result) they provided is not under their accredited scope and therefore follows the opinions and interpretations with it. In which you need to perform an assessment if this opinion and interpretation has an impact on your process
2. It is another way of informing you that the results provided are not under their Scope of Accreditation (this is as per agreement).
But in any case, this is not a problem as long as you completely understand the results of the report that they have issued as per your process usage and that you have agreed about the reported results.
As the user of the calibrated instruments, you must fully understand the results that are being reported in the calibration certificate. In any case, you cannot understand, you may ask the calibration lab for some explanations or interpretations of the results.
I hope this helps
Thanks for reading my post.
Edwin
Wahiba REMILI
Hello Edwin,
Thank you very much for your detailed explanation, much appreciated.
it helps me a lot to understand how to deal with laboratories, what scared me was the mention ‘out of the scope of work’, it is like the certificate is issued on his laboratory’s name and he says that what’s mentioned in the certificate is out of his scope of work something that worried me.
Again thanks a lot
edsponce
Hi Wahiba,
You are welcome.
There are 2 meanings of the term ‘out of the scope of work’, these may be:
1. They are capable to perform the calibration but they are not accredited to it, or
2. They cannot do the job because they do not have the capability to do it.
What I understand now is that, because it is ‘out of their scope of work’, they use another lab (we call this a 3rd party lab) to do the calibration for them, in your behalf, or in other words, they do not have the capability to do so that is why another lab performs the calibration for them.
This is a normal process, as long as the other lab (3rd party lab) is also an accredited lab that meets your requirements, I do not see any problem.
The only problem is that they should ask your permission or you should have an agreement about this.
I cannot give an exact answer since I did not see the certificate. If you want me to check it out so that I can give a more detailed comment, you can send it to my email, edwin@calibrationawareness.com.
Thanks and regards,
Edwin
Charlie
Sir Edwin, do you conduct seminars or trainings here in Philippines?
edsponce
Hi Charlie,
Sorry to inform you that I do not conduct training or seminars (yet), but I would be glad if I had a chance.
For now, this website is where I share my knowledge and experience.
Thank you for reading my post. If ever you have other related concerns, please feel free to comment further.
Best Regards,
Edwin
CECILE JOY
Good Day!
How often will you conduct the verification if you have already had the calibration interval of a certain equipment?
Thanks,
CECILE
edsponce
Hi Cecile,
The verification period is the same as the calibration interval. Verification is implemented every time a calibration is performed. I also conduct an intermediate check every 3 months where another verification is performed.
You need to continue the verification as per your normal interval to continuously monitor the performance of the instrument. This will help you to extend more the calibration interval or maintain the fixed interval chosen.
I hope this helps,
Edwin
Eduardo Sotomayor
What is the calculation that have you determine the Expanded Uncertainty for (1.01+/-0.007960)mV at a confidence level of 95%?
What is the process you use for it?
Thanks!
edsponce
Hi Eduardo,
We use the GUM (ISO/IEC 98-3:2008) approach in calculating the measurement uncertainty where Type A (repeatability) and Type B (resolution, drift, specifications/accuracy, standard uncertainty and others) contributors are combined to come up with final expanded uncertainty, K=2 at 95% confidence level.
I hope this helps.
Edwin
Madoda Nanto
Dear Sir
Please assist me with this question:
1. CMM calibration is done by an external company.
2. The techinician is qualified to the calibration
3. The bar used is also calibrated and traceable to the national standard
The only issue is that the company is doing the calibration is not accredited to the ISO 17025 standard.
Can the company continue or we should look somewhere else?
Thank you
edsponce
Hi Madoda,
An accredited laboratory should be the first to consider when using the service of an external lab. The requirement is to use a competent laboratory, where competency is evident if the lab is accredited.
But it is not always the case. You can also determine the competency of a lab by verifying (through an audit) that they follow the requirements of a quality system like ISO 17025.
First of all, check your policy regarding 3rd party calibration. Does it say you need to use an accredited lab? Or as long as you have taken the necessary control like verification for training, quality system documentation, and traceability is enough?
There are exemptions where not accredited labs can be used. These are:
1. No available accredited lab for the specific instruments(need to show evidence that you made a search)
2. The Instrument has a warranty from its manufacturer (OEM)
3. OEM is the only available lab that can provide the service.
4. You have performed an audit where the requirements set (like ISO 17025 requirements) are met and satisfied.
To avoid possible issues with the auditors and to ensure competency without an in-depth verification or audit, it is better to use an accredited lab.
I hope this helps.
Edwin
faheem haider
following example is wrong, do you agree
Example
Nominal value = 10.00
UUC reading = 9.24
Therefore 10.00 – 9.24= 0.276
edsponce
Hi Faheem,
You are right, there is a mistake, a good catch! I already corrected it.
It should be 10-9.924 = 0.076, which I based on the given table above.
Thanks for informing me. I appreciate the time you spent in reading my posts.
Best regards,
Edwin
edsponce
Hi Faheem,
Are you a calibration lab? If you are then you should choose the national standard for your traceability. The best traceability chain to be connected on should be 1 step ahead of you (at least 1 step higher in the traceability pyramid) in order to attain a good uncertainty value.
But if the uncertainty or CMC of the Local Standard is acceptable for you, then I believe it is not a problem.
If you are a 3rd party lab, sometimes this will become a problem if auditors or accreditation bodies will not accept it. Therefore, it is advisable to consult them if you are applying for accreditation.
I hope this helps,
Edwin
Carlos
Hi Edwin, I found your post really helpful. I’m new in this world of calibrations, so, I’ll really appreciate if you could clear my doubt.
what’s about the calibration factory cert for a new equipment (ex. a digital caliper) it is valid? if your answer is afirmative, what should be the start date of the calibration?.
thank you in advance.
regards.
edsponce
Hi Carlos,
I need more details on your question. Why do you ask for a start date of calibration, Is the equipment stored for a long time? Is the certificate a valid calibration certificate? But below are some of my inputs..
There are certificates that are issued by an OEM (Original Equipment Manufacturer) that is accredited under ISO 9001. These certificates are what we call traceable calibration certificates which are valid to use. It has a calibration date and due date that you can immediately follow, it is already written in the certificate.
Some certificate does not reflect due dates, it is up to the user to determine that.
Other OEM also put a remark in their certificate that the start date of calibration will be the date where it is newly open or installed. In the case of the caliper, if you can ensure that the caliper is not used after purchased and kept in a controlled room, you can use a calibration date as the day you start using it. But before using it, you need to perform a simple verification, if you have similar equipment that is calibrated, try to perform a comparison, or use a sample that has a good history of accepted results.
Some certificates are just a quality check report where only a pass or failed remarks is reflected in the report. If this is your case then it is not a valid calibration certificate.
I hope this helps.
Edwin
Carlos
Hi Edwin, thank you for your time and your help. Based on the info that you shared in your post, the OEM cert acomplish with all the info required. I asked you about the initial date because the cert don’t have one on it, but you already answer my question.
Do you think its necesary to add a copy of the equipment invoice with the OEM cert to validate the date?
regards.
edsponce
Hi Carlos,
It is good to know that your question is already answered.
Regarding the invoice, it is not necessary, the important is the record showing the date you start using the caliper. but it is still good to have it readily available.
But as part of quality control, a certificate with no calibration date should not be accepted, it is already a non-conformance, a cal-date is one of the main requirements in a calibration certificate and it is a part of document traceability. You may have an invoice date but we do not know how long it is stored on the supplier side before you purchased it. Therefore, it is better to have it recalibrate than to face an issue in the future.
I hope this helps,
Edwin
Carlos
Hi Edwin, sorry for my late response. Thank you for your help I really aprecciate it. You clarified all my doubts about this topic.
regards.
edsponce
Hi Carlos,
You are welcome. I am glad that your doubts were cleared.
Thanks and regards,
Edwin
kth
Know anything about acceptance criteria ?
How to judge whether the equipment after calibration is pass or fail by using acceptance criteria & uncertainty?
edsponce
Hi kth,
You are referring to a ‘decision rule’.
Based on the requirement of ISO 17025, if you need to judge whether the instrument has passed or fail based on tolerance limit or acceptance criteria, you need to account for the measurement uncertainty before making judgments or conformity assessment.
There are 3 ways in which you can do this:
1. Binary Decision Rule Simple Acceptance
2. Non-Binary Decision Rule
3. Guarded Decision Rule
The most used decision rule is the “Non-Binary Decision” rule wherein you need to include the measurement uncertainty in the measured value (MV) before making a decision (MV + uncertainty should be within the acceptance criteria). But since this rule has an indeterminate result, it will lead to the use of the “Guarded Decision Rule”.
Read my separate post in this link to better understand >> decision rules
Thanks for visiting my site,
Edwin.
Mohamed habashy
Thanks for your valuable information .
I need to ask a question if you don’t mind.
, Why sall do calibration I’m case of the results of CRM are within tolerance.
edsponce
Hi Mohamed,
Sorry I do not understand the question, kindly give more details.
Thanks,
Edwin
Thao Le
hi you , How to Calculate Correction Factor ?
edsponce
Hi Thao,
The correction factor is calculated simply by subtracting the Standard value and the UUT value, CF= STD-UUT. I have a full article for this, check out my other post in this link >> Correction Factor
Best regards,
Edwin
Jessie Holloway
Thanks so much for explaining how o read a calibration certificate. My brother has been talking about needing one done for his measuring instruments. I wanted to know more about what it was and how it’s used so I can help him look for companies that provide that service.
Thao Le
What section or part of iso17025 requires the application to calculate CF?
edsponce
Hi Thao,
Below is the section or clause related to using the “correction factor”.
see clause 6.4.11, which states that: When calibration and reference material data include reference values or correction factors, the laboratory shall ensure the reference values and correction factors are updated and implemented, as appropriate, to meet specified requirements.
Thanks and regards,
Edwin
RAHUL
hi
iam rahul
iam very new to calibration.
why beam gauges(used for measuring ID & OD) doesn’t have calibration
edsponce
Hi Rahul,
If you are using it for measuring ID and OD, it should be calibrated, unless there is a reason set by your quality department why calibration is not needed.
You can read about why calibration is not required in this link >> calibration not required
Best regards,
Edwin
Roberto
Dear Edwin,
Thanks a lot for this article on your blog. I am still quite new to the world of calibration and this explanation sure helps.
Recently I’ve had the pleasure of undergoing an audit for temperature controlled facilities to store pharmaceutical products. Indeed as you have written in this article, I’ve handed over the calibration certificates for our cold storages under the perception that this would be all fine.
The auditor however was shocked. Apparently the tolerance of 2 degrees Celsius would have been too much, as only a deviation of 0.5 degrees Celsius is apparently allowed. In addition he told me that would should have carried out a 3-point calibration for all the sensors present, with the lower, upper limits and the operating set point of the cold stores.
When I asked him where this was written in the regulations, he initially referred to the GDP and WHO regulations. However when I pointed out that GDP does not specifically mentioned the 3-point calibration or 0.5 degrees deviation limit, he stated that GDP refers to national and international regulations, hence referring to ISO 17025. Since then I am fascinated about ISO 17025, yet I still struggle to find the exact requirements as the auditor has pointed out.
I would appreciate your opinion and recommendations for further study on this topic.
edsponce
Hi Roberto,
Good day!
Communication with the calibration labs pointing out your requirements should be the first step. Therefore, it is important that you should know the requirements of your process prior to calibration.
ISO 17025 does not specify a specific requirement for a calibration procedure or methods. ISO 17025 is purely requirements and guidelines to perform and manage competent calibration or testing laboratories in terms of quality and technical competency.
A specific procedure and/or requirements come from the following:
1. Manufacturer recommendations
2. Internationally accepted guidelines like ASTM (ASTM E2500 – 07), Euramet, and DKD (Euramet cg-20 and DKD R 5-7 for enclosures)
3. Validated In-house calibration methods
4. Requirements from regulatory authority for Pharmaceutical Industries for a specific applications
5. Others
The above references and the needed tolerance limits or acceptable deviations should be communicated to the calibration lab prior to performing the calibration. You should discuss this with the calibration lab if they can perform the chosen procedure.
I hope this helps,
Edwin
Firdausul
Dear Sir,
just need information, is it possible calibration function name on cert same as authorize person?
i
edsponce
Hi Firdausul,
Yes, they are the same, but it should be clear that the same person is authorized to approve the certificate. He may be authorized to perform the calibration but not authorized to approve the certificate.
Thanks and regards,
Edwin
Faten
Very useful information I assure that every technician, students, calibrators and lab operators ….etc will understand the topics easily it is beneficial for me.
Thank you very much
edsponce
Hi Faten,
You’re welcome, I am glad it helps you.
Thank you for your comments as well.
have a nice day,
Edwin
Ashwini
Dear Sir ,
As per clause 7.8.8.3 under “Reporting of Result”, how to issue completely new report? how to uniquely identified and can report the certificate. Little confusing on understanding 7.8.8.2 and 7.8.8.3 on different aspects.
edsponce
Hi Ashwini,
The clause falls under “Amendments to Report”.
The clause requires you to document any changes when releasing or issuing a new calibration report in case you need to change or revise an old one.
7.8.8.2 requires to have a further document before issuing a new calibration report. You need to have a form that you need to accomplish which contains the necessary information and approval before a new calibration report is issued. You can call it an “Amendment to Report Form” or “Change Request Form”, it’s up to you. The main purpose is to have an approval process before the amendment of a calibration report takes place.
And clause 7.8.8.3, requires you to uniquely identify the amended calibration report but with a reference to the old one it replaces. For example, the certificate number of the original certificate is CR123456, now the amended or newly issued report will now become CR123456-A, (A) is the unique identifier. Do not forget to document this in the form you accomplish above.
I hope this helps,
Edwin
Naomi Allkins
Good Afternoon Edwin,
I thought this would be a great place to put feedback on the training you so kindly put together for our company on this subject, as it was through this article I reached out to you.
The training you provided on this subject was very informative, clear and concise, I certainly got a good refresher on my knowledge and it really helped out team to better understand also. I have been given great feedback from the attendees.
Thank you Edwin, for sharing your knowledge.
edsponce
Dear Naomi,
Good day! You’re very welcome. It is my pleasure to share my knowledge with you, thank you as well for choosing me to provide this training, I am glad that it provides a good refresher and understanding in this subject.
Thank you also for this heart warming feedback, I appreciate it very much. This motivates me more to share and create more content that can help others as well.
Hope to connect with you again. Have a nice day!
Best regards,
Edwin
Nobu T.
Hi Sir,
May I ask if we could compute the correction factor from the Nominal Value and As Left values? This is from the manufacturer’s issued calibration certificate. Thank you.
edsponce
Hi Nobu,
Correction Factors can only be computed once you have known the reference value. Please note that the formula to calculate correction is “STD Value-UUT Value”. Therefore, you cannot calculate correction factor from the nominal and As left values. Try to ask the manufacturer if the nominal value is considered as the reference value or the value from the reference std they used.
Thanks and regards,
Edwin
HALA
WHAT IS THE TOLERANCE LIMIT?HOW TO KNOW IT ?
edsponce
HI Hala,
Please visit my other post to answer your questions. This is the link >> Tolerance limit
Best regards,
Edwin
Carlos
Hi.
I have a question about measurement uncertainty. I have received a calib cert of an RTD and it reads somewhat like this. Btw I am a product test engineer.
Ref std= 27.00degC UUC indication=27.00degC Error= 0.00degC Expanded U=0.29degC
Ref std= 28.00degC. UUC indication= 27.98degC. Error=-0.02degC. Expanded U=0.29degC
Ref std=29.00degC. UUC indication=29.02degC. Error=0.02degC. Expanded U=0.29degC
1. How come that there is no error on the equipment, but the calib lab still reported an MU?
2. Also, for example RTD is used for testing under a certain product standard, And it requires that the expanded MU of temperature parameter shall be at 0.20degC only. Does it means that the RTD is now out of tolerance as it failed to meet the requirement of the product standard?
Please enlighten me on this. Thank you.
edsponce
Hi Carlos,
Below are the answer to your queries:
1.Measurement uncertainty is for the UUC indication or measured value, not for the error. Remember that there are no exact results, there is always an error that we cannot see (doubt), and this is where the measurement uncertainty comes in, to quantify the doubt. This means that the exact value is within 27.00+/-0.29, same thing with the other measured value.
2.0.20 deg C is a requirement for measurement uncertainty, not a tolerance limit. If the measured value +/- MU exceeds the tolerance limit, that is where it becomes out-of tolerance. Since you did not meet the specified MU requirements, you only need to look for a lab that can meet that requirement or choose an RTD (measuring instrument) with better accuracy.
I hope this helps. Thanks for visiting my site.
Best regards,
Edwin